 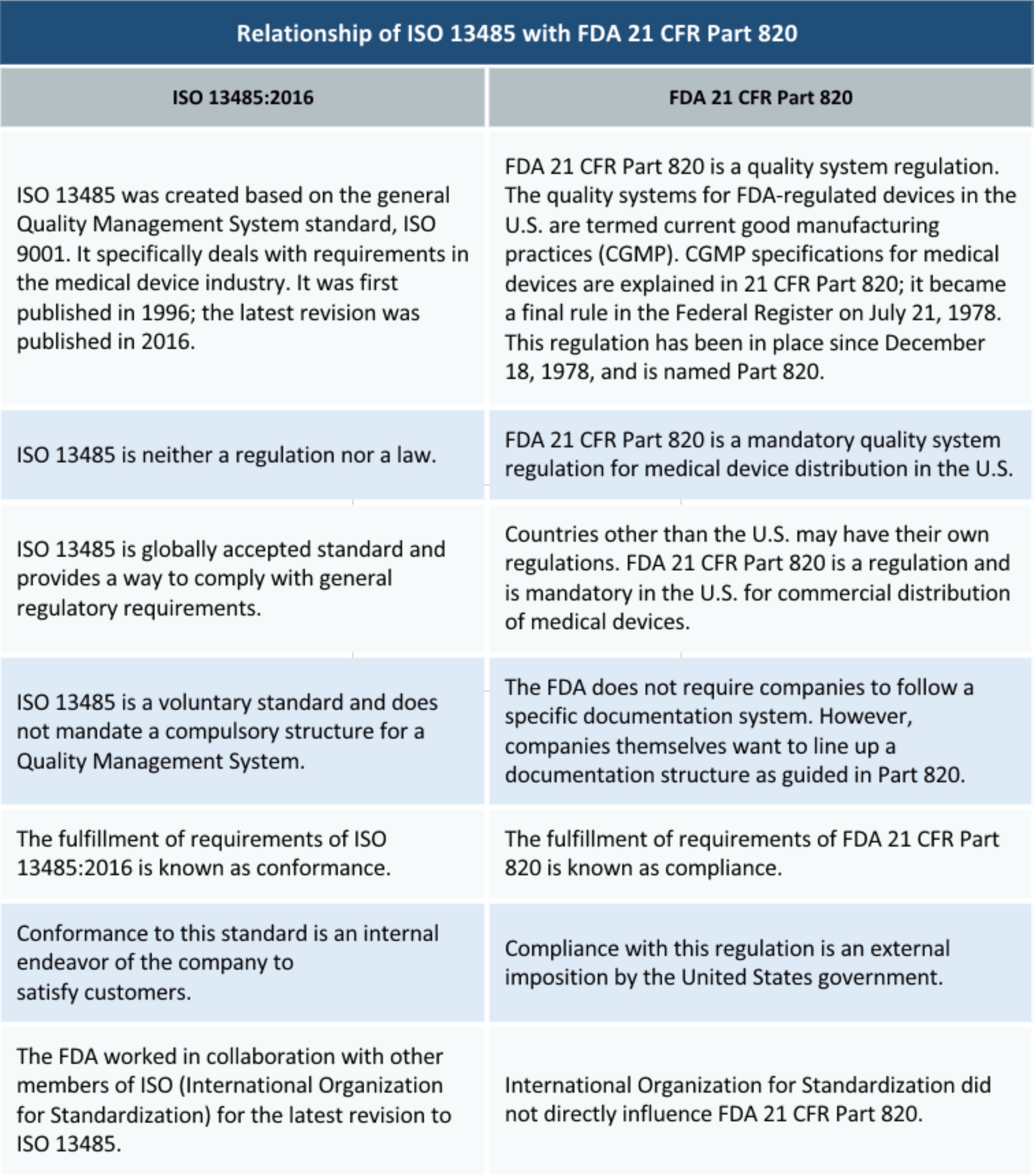
ISO 13485 represents the requirements for comprehensive organizational goals and aspirations, policies, processes, documented information, and resources needed to implement and maintain this standard or a quality management system for the design and manufacture of medical devices, from simple devices (e.g., tongue depressors, thermometers, stethoscopes, blood pressure cuffs, latex gloves) to complex devices (e.g., computer-assisted medical testing, cochlear ear implants, prostheses). International Organization for Standardization (ISO) is an independent, non-governmental, international organization with a membership of 163 national bodies (including the United States) that set class specifications for products, services, and systems to ensure quality, safety, and efficiency. Stanley ( is an Associate in the Lexington, KY office of Quintairos, Prieto, Wood & Boyer, PA.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
